Monoclonal antibodies (mAbs) are being discovered and developed as new therapeutic approaches to combat different diseases such as cancers, autoimmune diseases, HIV and more. Some immunoglobulin G (IgG) mAbs have been shown to affect platelet function with risk of thrombotic or bleeding side effects occurring in patients. They can induce platelet activation by different mechanisms via the FcgRIIA receptor expressed on platelets, known to have low affinity for the constant fragment (Fc) of IgG. Platelet function testing can provide a useful tool for companies developing therapeutic antibodies to investigate the possible effects of their mAbs on platelet function early in a project to identify potential risks of either bleeding or thrombosis.
Platelet activation via cross-linking of FcgRIIA receptors
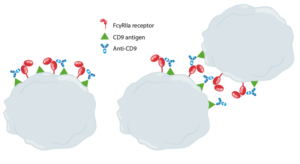
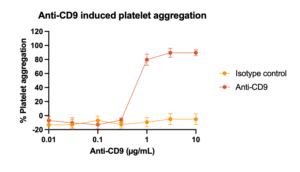
Cross-linking of FcgRIIA receptors, in clusters on the same or adjacent platelets, by mAbs leads to platelet activation. If the antibody target is expressed on platelets, the Fab portion of mAbs binds to their target antigen and the Fc portion binds to the FcgRIIA receptors causing cross-linking of FcgRIIA receptors. For example, an anti-CD9 (an IgG1 subclass), was shown to induce platelet activation by this mechanism.
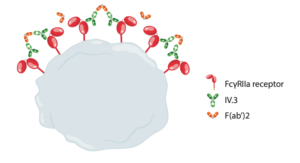
Antibody interactions can be used to dissect out/confirm the mechanism(s) underlying antibody-induced platelet activation. For example, IgG mAbs targeting FcgRIIA (one common clone is IV.3) can inhibit platelet activation by preventing the binding to FcgRIIA of antibodies that could cause platelet activation (as described in the scenario above) and can be used as an in vitro tool to confirm Fc-mediated nature of any possible effects of the antibodies. Conversely, cross-linking of IV.3 antibody with F(ab’2) fragments of a secondary antibody can also induce FcgRIIA clustering and lead to platelet activation.
Platelet activation via immune complexes
Immune complexes containing clusters of IgG have been reported to activate platelets via FcgRIIA receptors. An example is ruplizumab (BG9588), an IgG1 antibody against CD40L, where clinical trials for the treatment of systemic lupus erythematosus were halted due to thromboembolic events occurring in patients. Once investigated, platelet testing studies in vitro showed immune complexes containing anti-CD40L mAbs and soluble CD40L (sCD40L) activating platelets through FcgRIIA. Soluble CD40L is a cleaved form of CD40L present on activated T-cells, macrophages, and endothelial ells but the major source of sCD40L comes from activated platelets.It is possible that sCD40L cleaved from platelets upon stimulation mediates the formation of anti-CD40L and sCD40L immune complexes that in turn activate platelets.
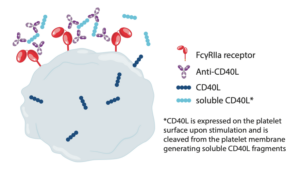
Due to the thrombotic events observed with ruplizumab, an anti-CD40L IgG lacking a functional Fc region, known as dapirolizumab pegol (DZP) (CDP7567), was developed. Studies in vivo with DZP demonstrated no thrombotic events in Rhesus monkeys and DZP failed to activate platelets in vitro. Dapirolizumab pegol is currently in phase III clinical trials and during phases I and II no serious thrombotic events occurred in patients.
Platelet function testing
Platelet Services offers an array of platelet testing assays from the “gold standard” light transmission aggregometry to higher throughput plate-based assays measuring either platelet activation or aggregation. These assays can be performed in three different matrices, whole blood (most physiological), platelet rich plasma and isolated (“washed”) platelets. Our team has extensive experience working with various mAbs and can design a study to efficiently determine the possible effects of mAbs on platelet function. Importantly, we have developed protocols in which the platelet activating effects of positive control/reference antibodies can be reliably and reproducibly demonstrated, providing a robust platform for identifying test antibodies that lack platelet activating properties.

